Integrating FcRn and Complement System Therapies for Generalized Myasthenia Gravis
Complimentary
Enduring Webcast
Released on 4/1/26
Expires on 4/1/27
Take Me to the Event Registration
Program Description
This enduring webcast addresses the autoimmune pathogenesis of gMG and the integration of targeted immune-mediated therapies. The activity will focus on:
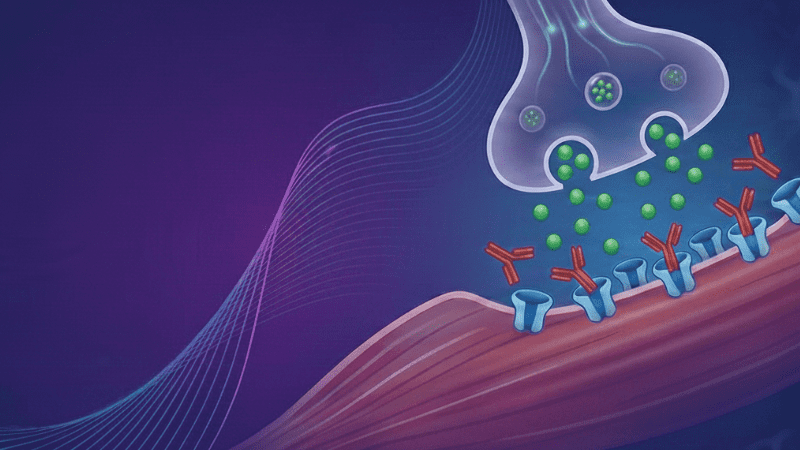
Autoimmune pathogenesis of gMG – Autoimmune pathology in gMG disease subtypes is governed by divergent mechanisms of immunopathology
- Anti-AChR autoantibodies – ~85% of gMG cases
- Anti-MuSK autoantibodies – 5-10% of gMG cases
- Anti-LRP4 antibodies
- Anti-agrin autoantibodies
- Seronegative gMG
Etiology and Classification – gMG occurs in genetically susceptible individuals, and precipitating factors include infections, immunization, surgeries, and drugs.
- Early-onset gMG: Age at onset less than 50 years with thymic hyperplasia
- Late-onset gMG: Age at onset greater than 50 years with thymic atrophy
- Thymoma-associated gMG
- The gMG with anti-MuSK antibodies
- Ocular gMG: Symptoms only from periocular muscles
- The gMG with no detectable AChR and MuSK antibodies
Pathophysiology – The pathophysiologic mechanisms in gMG depend on the type of antibodies present.
- Nicotinic acetylcholine receptors (n-AChR) gMG
- Muscle-specific kinase (MuSK) gMG
- Lipoprotein-related protein 4 (LPR4) gMG
- Clinical classification of gMG
- The Myasthenia Gravis Foundation of America (MGFA) clinical classification divides gMG into 5 main classes based on the clinical features and the disease severity.
Evaluation – The diagnosis of gMG is primarily clinical. Laboratory investigations and procedures usually aid clinicians in confirming clinical findings.
- Serologic Tests
- Electrophysiologic Tests
- Repetitive nerve stimulation (RNS) test
- Single-fiber electromyography (SFEMG) test
- Edrophonium (Tensilon) test
- Ice-pack test
- Imaging: Chest computed tomography (CT) or magnetic resonance imaging (MRI)
Treatment/Management of gMG
- Symptomatic treatment
- Immunosuppressive treatment
- Intravenous immunoglobulins (IVIG)/plasmapheresis
- Thymectomy
Target-specific Therapies
- FDA-approved treatments for gMG include:
- FcRn Inhibitors – Efgartigimod, Rozanolixizumab
- Complement System Inhibitors – Eculizumab, Zilucoplan, and Ravulizumab
- Ongoing clinical trials
- Nipocalimab – The mAb targeting FcRn
- Batoclimab – The mAb targeting FcRn
Intended Audience
Neurologists, neuromuscular specialists, ophthalmologists, primary care physicians, advanced practice providers (physician assistants and nurse practitioners), and nurses who manage patients with gMG.
Commercial Supporter
Supported by an educational grant from UCB Inc.
Learning Objectives
Upon completion of the educational activity, participants should be able to:
- Summarize the etiology, classification, pathogenesis, and evaluation of generalized myasthenia gravis (gMG).
- Apply symptomatic, immunosuppressive, immunoglobulin/plasmapheresis, FcRn, and complement system therapies for gMG.
Accredited Providers
Jointly Accredited by Amedco and MedNet


Joint Accreditation Statement
 In support of improving patient care, this activity has been planned and implemented by Amedco and MedNet. Amedco is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
In support of improving patient care, this activity has been planned and implemented by Amedco and MedNet. Amedco is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Physicians (ACCME) Credit Designation
Amedco designates this live activity for a maximum of 1.25 AMA PRA Category 1 CreditsTM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Physician Assistants
PAs may claim a maximum of 1.25 Category 1 credits for completing this activity. NCCPA accepts AMA PRA Category 1 CreditsTM from organizations accredited by ACCME or a recognized state medical society.
Nurse Practitioners
The American Association of Nurse Practitioners (AANP) recognizes the Accreditation Council for Continuing Medical Education (ACCME) and the American Nurses Credentialing Center (ANCC) as approved accreditors and allows reciprocity for AANPCP continuing education credit. 1.25 hours.
Nurses
This activity is awarded 1.25 contact hours (based on 60 minutes per contact hour).
Faculty

Jonathan Morena, DO
Assistant Professor of Neurology
Division of Neuromuscular Medicine
Duke University
Dr. Morena discloses the following relevant financial relationships with ineligible companies:
Consultant: Adivo Associates
The following relationships have ended within the last 24 months:
Consultant: Amgen, Optio Biopharma Solutions
Advisory Board: Johnson and Johnson

Karissa Gable, MD, FAAN
Professor of Neurology
Division of Neuromuscular Medicine
Neuromuscular Fellowship Director
Neurology Medical Student Clerkship Director
Duke University
Dr. Gable discloses the following relevant financial relationships with ineligible companies:
Consultant/Adjudication Committee: Argenx, Dianthus Therapeutics, Immunovant, Inc., and Sanofi
Consultant/PI: Takeda (TAK-881 study)
All of the relevant financial relationships listed for these individuals have been mitigated.
Instructions
In order to receive CME/CE credits, participants must complete the pre-assessment questions, post-assessment, and program evaluation. Participants must also score at least 75% on the post-assessment. Certificates will be distributed online at the conclusion of the activity. Your online certificate will be saved on myCME within your Dashboard or Transcript, which you can access at any time.
Related products
-

New Prospects in Blood-based Biomarkers and Disease-Modifying Anti-Amyloid Antibody Therapies for Early-Stage Alzheimer’s Disease
Read more -

Glaucoma: Sustained-release Drug Delivery System, Topical Agents, and Emerging 3D Visualization Technologies
Read more -

New Paradigms in the Treatment and Management of Age-related Macular Degeneration (AMD)
Read more -

Glaucoma Management: Understanding Risk Factors, Screening and Treatment Options
Read more